Seesaw molecular geometry example8/19/2023  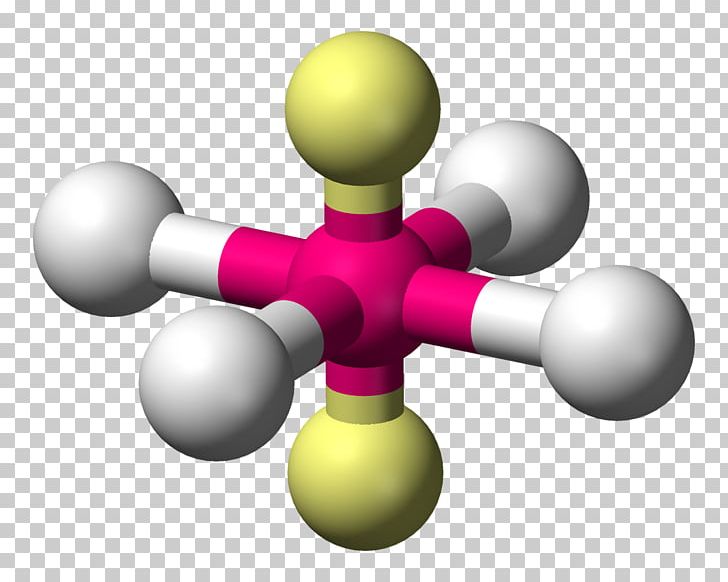
So, that bends the axial fluorines together a bit. Top Rohan Kubba Dis 4B Posts: 50 Joined: Sat 7:18 am Re: seesaw Postby Rohan Kubba Dis 4B » Sat 5:10 am Essentially this is when there are 4 bonds to a central atom and a lone pair. Note though that the structure is distorted a bit due to the repulsive forces of the lone pair of electrons you see (not bonded). A molecule with a seesaw molecular geometry means that the molecule has four bonding pairs and one lone pair. With one lone pair of valence electrons, you get a seesaw molecular geometry. With the bridge in place, the team could now take the molecular tile and work upward to constrain its potential large-scale patterns using a simple system of five algebraic equations and inequalities that could. 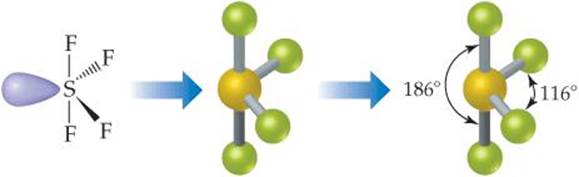
Therefore, you can put 6x4 on each fluorine, 2x4 to account for four single bonds, and 2 for the last 2 valence electrons available.Īs a result, you have 5 electron groups, so the electron geometry would be trigonal bipyramidal. The equation describing this bridge linked the geometry of the smallest and middle levels with the largest level, the molecular mosaic. You can put sulfur in the middle because fluorine tends to make single bonds. Hope this helped.Within the context of VSEPR theory, you can count electrons to determine the electron geometry ("parent" geometry). The nitrogen has 5 valence electrons and thus needs 3 more electrons from 3 hydrogen atoms to complete its octet. It's helpful to draw out what the structures would look like if there was a bonded atom in place of the lone pair for visualization purposes. It is perhaps less obvious that the shape of a molecule may. Trigonal Planar Molecular Geometry VSEPR An example of trigonal pyramid molecular geometry that results from tetrahedral electron pair geometry is NH 3. The sulfur atom is bonded to four fluorine atoms (4 areas of electron density) and has one lone pair (1 area of electron density). There are five areas of electron density (compared to four areas of electron density in SO 3 2-). 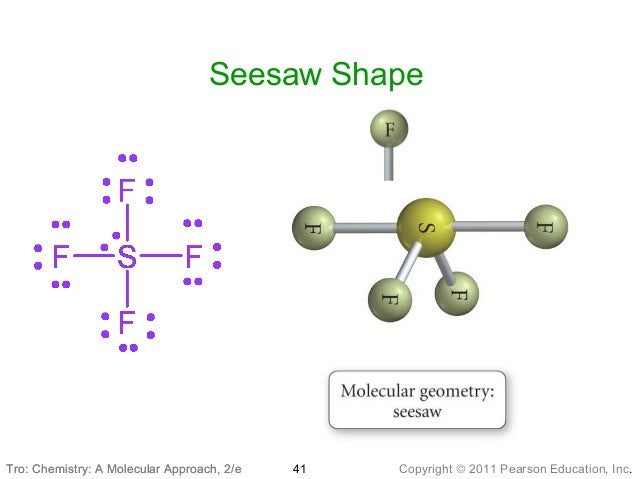
(b) The trigonal pyramidal molecular structure. In contrast, SF 4 has five atoms involved in its structure: a sulfur atom and four oxygens. (a) The electron-pair geometry for the ammonia molecule is tetrahedral with one lone pair and three single bonds. Trigonal bipyramidal also has five areas of electron density but all five are bonded pairs. **Edit: Sorry I read trigonal bipyramidal as trigonal pyramidal. Examples Sulfur tetrafluoride is the premier example of a molecule with the disphenoidal molecular geometry (see image at upper right). There are only four atoms involved in this structure: sulfur and three oxygen atoms. For example, if we want to obtain the Lewis structure of the Sulfate ion. This gives it the shape of trigonal pyramidal. It forms a see-saw shape and has a trigonal bipyramidal molecular geometry. So that gives it a total of four areas of electron density. An example of an octahedral molecule (AX 6) is sulfur hexafluoride (SF 6). molecular geometries in this domain: Trigonal bipyramidal Seesaw T-shaped Linear. The sulfur atom is connected to three oxygen atoms (3 areas of electron density) and then it has a lone pair (1 area of electron density). With one lone pair on this electronic geometry, the lone pair must occupy an equatorial position and the molecular geometry becomes see-saw shape. This is the Valence-Shell Electron-Pair Repulsion (VSEPR) model. The trigonal pyramidal structure actually has four areas of electron density. 
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
